-
Name:Carfilzomib
Catalog No.:B0016
Structure: 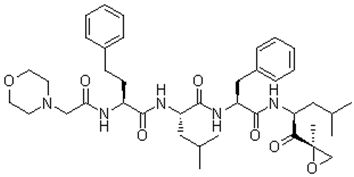
Chemical Properties Cas No.:868540-17-4 Purity Limit:99% Synonyms: Molecular Weight:719.91 Formula:C40H57N5O7 Storage: Solubility:
Shipping Condition
Quality Control & MSDS
Description Carfilzomib, also known as PR-171 is a tetrapeptide epoxyketone and an epoxomicin derivate with potential antineoplastic activity. Carfilzomib irreversibly binds to and inhibits the chymotrypsin-like activity of the 20S proteasome, an enzyme responsible for degrading a large variety of cellular proteins. Inhibition of proteasome-mediated proteolysis results in an accumulation of polyubiquinated proteins, which may lead to cell cycle arrest, induction of apoptosis, and inhibition of tumor growth. Check for active clinical trials or closed clinical trials using this agent. (NCI Thesaurus). Carfilzomib was approved by the FDA for use in patients with relapsed and refractory multiple myeloma on 20 July 2012. Onyx expects to launch the drug in the U.S. on 1 August 2012. When it launches, it will cost $10,000 per 28-day cycle, making it the most expensive FDA-approved drug for multiple myeloma
References Related products
- Talabostat
Tel: +86-411-39042497
sales@alphabiopham.com
Contact us now!
We'll offer you the best prices
 登录 | 注册
登录 | 注册 sales@alphabiopham.com
sales@alphabiopham.com 4000707518
4000707518

